Lewis Dot Structure
Lewis Structures are important to learn because they help us predict: the shape of a molecule. How the molecule might react with other molecules. The physical properties of the molecule (like boiling point, surface tension, etc.). A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom.
- Lewis structures are structural formulas for molecules and polyatomic ions that represent all valence electrons. Since valence electrons are typically represented as dots, these structural formulas sometimes are called Lewis dot stutctures. Here we present some examples of how to draw Lewis structures.
- Draw the Lewis dot structure for element. To change the symbol of an atom, double-click on the atom and enter the letter of the new atom. Periodic Table of Elements Element Gallium - Ga. Comprehensive data on the chemical element Gallium is provided on this page; including scores of properties, element names in many languages, most known.
- FREE Expert Solution. We're being asked to identify the incorrect Lewis structure. For (1):. Nitrogen is from Group 5A so it must have 5 valence e– around it. It also prefers to follow the octet rule. N: valence electrons = 1 lone pair (2 e –) + 3 bonds (1 e –) = 5 e –. N: total electrons = 1 lone pair (2 e –) + 3 bonds (2 e.
Learning Objectives
By the end of this section, you will be able to:
- Write Lewis symbols for neutral atoms and ions
Lewis Symbols of Monoatomic Elements
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful.
A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side. (It does not matter what order the positions are used.)
For example, the Lewis electron dot diagram for calcium is simply

Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table.
Figure 1. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table.
Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium:Likewise, they can be used to show the formation of anions from atoms, as shown below for chlorine and sulfur:Figure 2 demonstrates the use of Lewis symbols to show the transfer of electrons during the formation of ionic compounds.Figure 2. Cations are formed when atoms lose electrons, represented by fewer Lewis dots, whereas anions are formed by atoms gaining electrons. The total number of electrons does not change.
Example 1: Writing Lewis DoT SYmbols of Elements
What is the Lewis electron dot diagram for each element?
- aluminum
- selenium
The valence electron configuration for aluminum is 3s23p1. So it would have three dots around the symbol for aluminum, two of them paired to represent the 3s electrons (or three single dots around the atom):
The valence electron configuration for selenium is 4s24p4. In the highest-numbered shell, the n = 4 shell, there are six electrons. Its electron dot diagram is as follows:
Check Your Learning
What is the Lewis electron dot diagram for each element?
- phosphorus
- argon
Example 2: Writing Lewis DoT SYmbols of Ions
What is the Lewis electron dot diagram for each ion?
- Ca2+
- O2−
Having lost its two original valence electrons, the Lewis electron dot diagram is just Ca2+.
Ca2+
The O2− ion has gained two electrons in its valence shell, so its Lewis electron dot diagram is as follows:
Check Your Learning
The valence electron configuration of thallium, whose symbol is Tl, is 6s25d106p1. What is the Lewis electron dot diagram for the Tl+ ion?
Key Takeaways
- Lewis electron dot diagrams use dots to represent valence electrons around an atomic symbol.
- Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions) dots than the corresponding atom.
Exercises
1. Explain why the first two dots in a Lewis electron dot diagram are drawn on the same side of the atomic symbol.
2. Is it necessary for the first dot around an atomic symbol to go on a particular side of the atomic symbol?
3. What column of the periodic table has Lewis electron dot diagrams with two electrons?
4. What column of the periodic table has Lewis electron dot diagrams that have six electrons in them?
5. Draw the Lewis electron dot diagram for each element.
a) strontium
b) silicon
6. Draw the Lewis electron dot diagram for each element.
a) krypton
b) sulfur
7. Draw the Lewis electron dot diagram for each element.

a) titanium
b) phosphorus
8. Draw the Lewis electron dot diagram for each element.
a) bromine
b) gallium
9. Draw the Lewis electron dot diagram for each ion.
a) Mg2+
b) S2−
10. Draw the Lewis electron dot diagram for each ion.
a) In+
b) Br−

11. Draw the Lewis electron dot diagram for each ion.
a) Fe2+
b) N3−
12. Draw the Lewis electron dot diagram for each ion.
a) H+
b) H−
Show Select Answer
1. The first two electrons in a valence shell are s electrons, which are paired.
3. The second column of the periodic table
5.
a)
b)
Lewis Dot Structure For Oxygen
7.
a)
b)
9.
a) Mg2+
b)
11.
a) Fe2+
b)
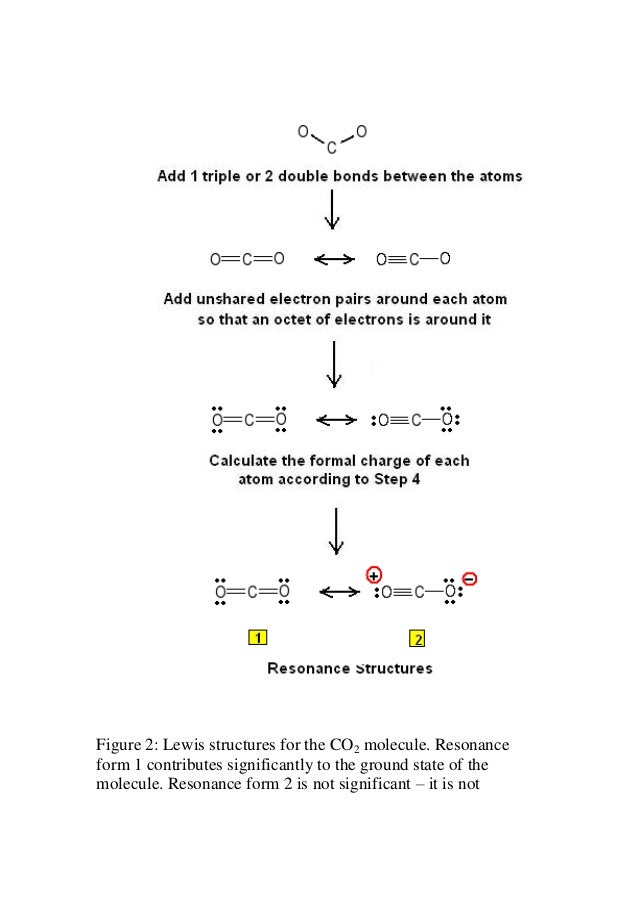
Lewis Dot Structure For Co2
A chemical element is identified by the number of protons in its nucleus, and it must collect an equal number of electrons if it is to be electrically neutral. As electrons are added, they fill electron shells in an order determined by which configuration will give the lowest possible energy. The first shell (n=1) can have only 2 electrons, so that shell is filled in helium, the first noble gas. In the periodic table, the elements are placed in 'periods' and arranged left to right in the order of filling of electrons in the outer shell. So hydrogen and helium complete the first period.
Lewis Dot Structure For Nh3
The number of electrons in a given shell can be predicted from the quantum numbers associated with that shell along with the Pauli exclusion principle. The second shell, associated with principal quantum number n=2, can have a maximum of 8 electrons and corresponds to the second period of the periodic table. The third shell also has 8 electrons, but things get more complicated after than because the subshells spread out enough that there is overlap between them.
Lewis Dot Structure 3d
| Lewis Dot Diagrams | Visualization of electron orbitals |